 Anode half reaction: Cathode half reaction: Shorthand. The electrode on the left is the anode, the one on the right is the cathode. Zinc is higher up on series than copper thus it is more easily oxidized. 100 (15 ratings) Transcribed image text: Complete the half reactions for the cell shown here, and show the correct shorthand notation for the cell by dragging the labels into the correct position. To determine which metal will be oxidized, or which will be reduced, refer to the activity series.The high concentration allows ions to diffuse out of it. Salt bridge contains a concentrated solution of a strong electrolyte.This is the voltage generated when two different solutions come into contact with each other Reduces the liquid-junction potential.Provides electrical continuity for anions and cations. 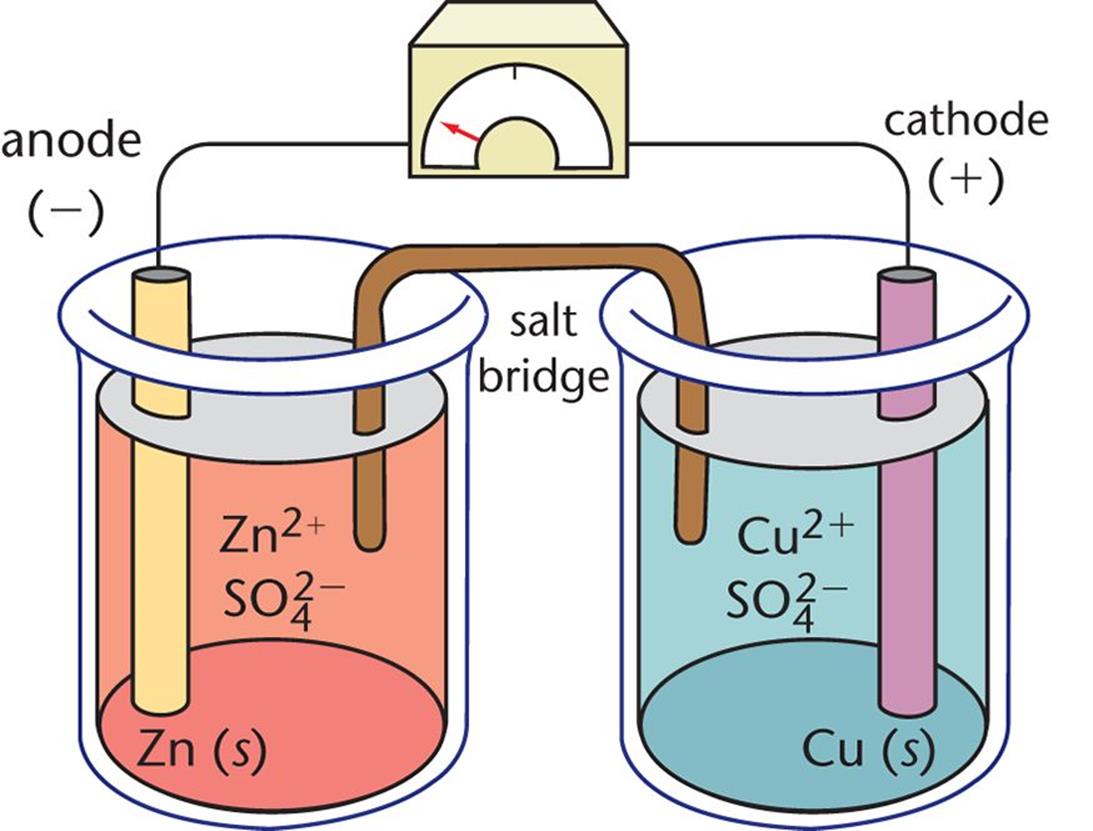
Provides physical separation of reduction and oxidation processes.Salt bridge which connects both electrodes has multiple functions:.The vertical line represents a phase boundary (junction).Consists of a bar of metal placed in a solution containing cations of the same metal.There are different types of electrodes used in voltaic cells, most common one is metal/metal ion electrode.Oxidation occurs at one half-cell (anode), reduction occurs at on half-cell (cathode) These two are separated, connected only by a salt bride. The salt bridge is represented by two parallel lines By convention, anode is always of left, and cathode on right. Cell Diagrams are used as shorthand notation to represent a voltaic cell.In both voltaic and electrolytic cells:.Electrochemical cells contain two electrodes, anode and the cathode.An electrode is a conductor of electricity used to make contact with a non-metallic part of a circuit, such as the solution in a cell (electrolyte).Electrolytic Cells – convert electrical to chemical energy.Reactions are spontaneous and exothermic. Voltaic (Galvanic) Cells – convert chemical to electrical energy.There are two main types of electrochemical cells.In an electrochemical cell, there can either be a conversion of chemical energy to electrical energy, or the other way around.D.7 (HL) Taxol – a chiral auxiliary case study.D.6 Environmental impact of some medications.D.1 Pharmaceutical products and drug action. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
